Capabilities
Key Features
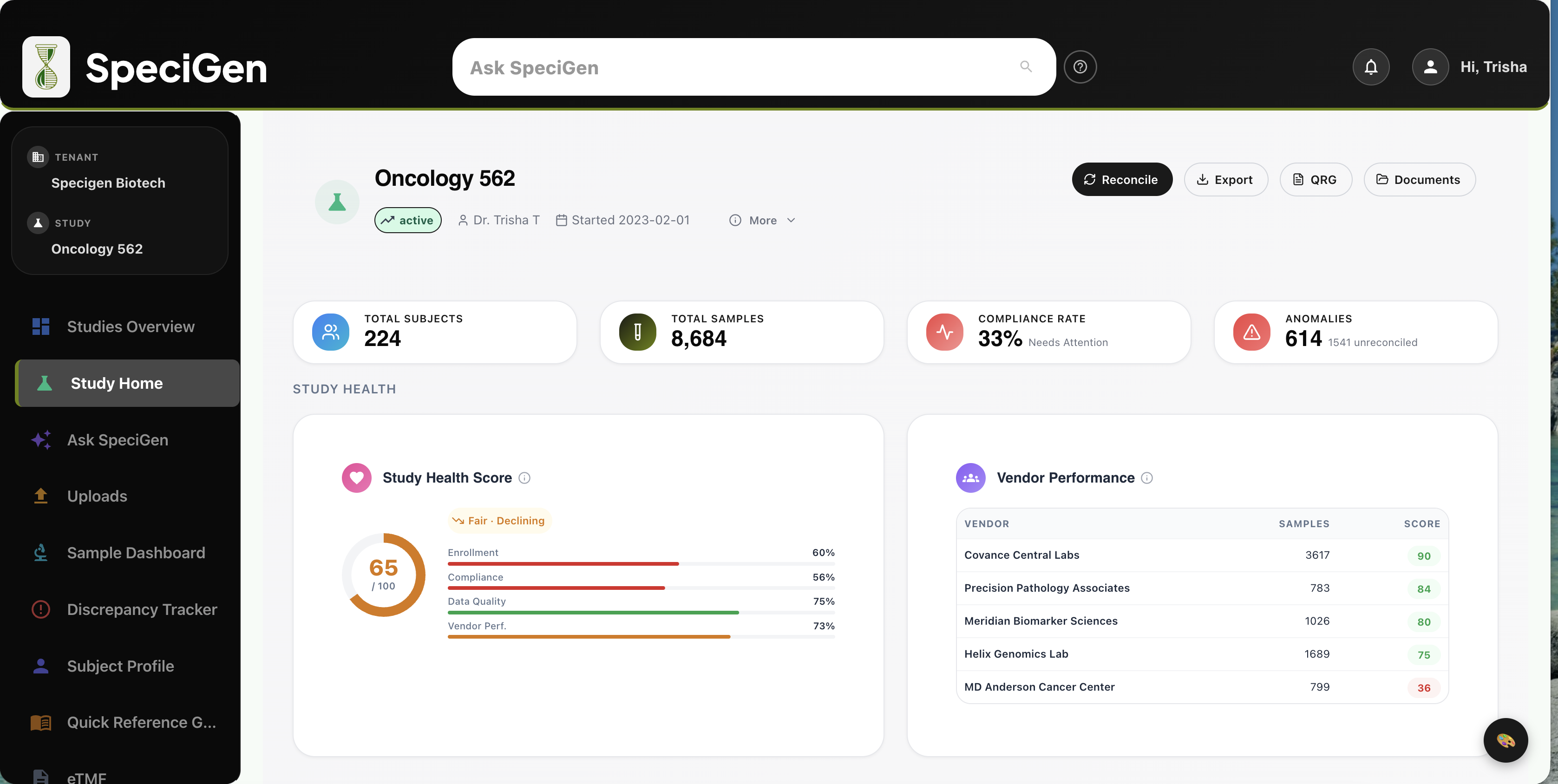
Everything you need to manage biospecimens across your clinical trials
Biospecimen Tracker
HubTrack biospecimen lifecycle across collection, processing, storage, shipment, and destruction.
Data Reconciliation
HubAuto-match lab samples to EDC records with manual override and anomaly detection.
Compliance & Audit
Hub21 CFR Part 11 compliant with e-signatures, consent tracking, audit logs, and study closeout.
Integrations & Security
HubEDC integration (Medidata Rave, Veeva, REDCap), analytics (Tableau, Spotfire), SSO, and encryption.

eTMF Generation
HubGenerate Clinical Study Reports and FDA-ready eTMF documents automatically.
Smart Annotations
HubExtract, annotate, and map data from PDFs — pathology and lab reports.
Results Tracker
LensIngest bioanalytical and assay results, trace samples to outcomes, and manage unblinded data with granular access controls.
Smart Insights
LensNatural language queries, real-time dashboards, and intelligent analytics for study metrics.